MRI Contract Agent Risk of Gadolinium Deposits in Brain Reviewed by EMA
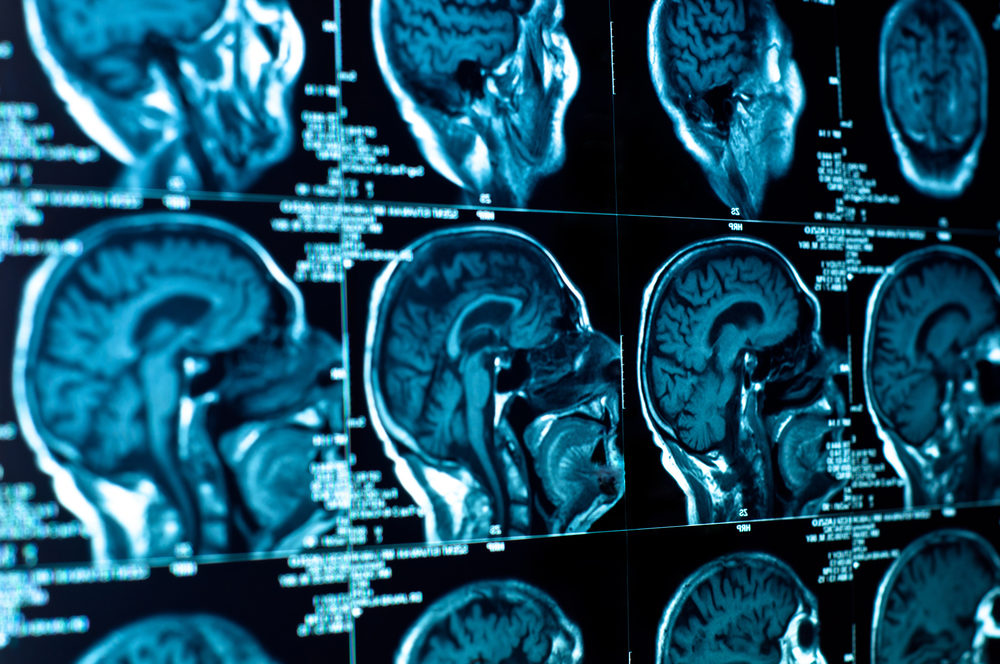
European health officials are investigating the risk that some MRI contrast agents may leave potentially toxic deposits in the brain.
Following a number of studies, and a similar investigation launched by the FDA, the European Medicines Agency (EMA) is examining the risk of gadolinium deposits on the brain. The investigation was announced in a press release (PDF) issued last month, and will be conducted by the EMA’s Pharmacovigilance Risk Assessment Committee (PRAC).
Gadolinium based MRI contrast agents are used to enhance the images and allow physicians interpreting the exam to distinguish blood vessels from nearby tissue.

Did You Know?
Millions of Philips CPAP Machines Recalled
Philips DreamStation, CPAP and BiPAP machines sold in recent years may pose a risk of cancer, lung damage and other injuries.
Learn MoreThe FDA launched its own review of the MRI contrast agent risks in July 2015, following a number of studies that suggested individuals undergoing the examination may experience a buildup of gadolinium on the brain. The FDA, and the EMA, are reviewing whether those deposits are harmful.
Two recent studies found that such gadolinium deposits occur, usually with multiple MRI examinations with contrast.
A year ago, a study published in the medical journal Radiology examined the brains of cadavers and found that those who had received gadolinium injections had deposits in the brain, while those who had not received such injections did not.
In June 2015, a study by European researchers in the journal Investigative Radiology found similar deposits in the brains of rats injected with Omniscan, a GBCA (gadolinium-based contrast agent) made by GE Healthcare. The rats developed significant, persistent MRI abnormalities over time, and high levels of gadolinium were found deep in the rats’ brains.
The FDA says that it will use its National Center for Toxicological Research to study the potential safety risks further, but is currently taking no action.
The PRAC reviewed the studies in January and will send its recommendations to the EMA’s Committee for Medicinal Products for Human Use. At this time, however, the PRAC reports that there have been no adverse effects reported in connection to gadolinium deposits in the brain.
Gadolinium Contrast Safety Concerns
Although the MRI dyes are generally believed to be safe, several years ago concerns emerged about a link between MRI gadolinium contrast agents and a severe and life-threatening skin condition, known as nephrogenic systemic fibrosis (NSF), which was found to occur among patients with impaired kidney function, causing their skin to thicken and harden, severely restricting movement.
Sometimes referred to as gadolinium associated systemic fibrosis, NSF is a painful disorder that has no known cure and often results in confinement to a wheelchair and then death.
In 2007, the FDA limited gadolinium contrast doses in most patients and contraindicated it for others, which limited the risk of NSF. In September 2010, the FDA went even further and banned the use of Bayer’s Magnevist on patients with kidney problems, due to the heightened risk of NSF.
The agency also required label changes for all GBCAs, warning healthcare professionals to screen patients before injecting gadolinium to identify those suffering from acute kidney injury or chronic, severe kidney disease.
Get more articles like this sent directly to your inbox.
"*" indicates required fields



0 Comments